Shape of My Cell: Reading the morphological "drift" of the cellular factory
As the promise of advanced cell therapeutics are increasingly recognised, potentially transforming medicine, the need for new innovation and scale up of successful solutions will demand increasingly accurate and reproducible measurements to ensure consistency and support the delivery of safe, efficacious and trusted solutions. As the designated institute for biological measurement, the UK’s National Measurement Laboratory (NML) at LGC is focused on meeting this challenge by advancing biotechnology through the development of methods to ensure greater accuracy and confidence in the analytical procedures necessary to develop and apply cell therapeutics. Jeanne Rivera, Science Area Lead for the Cell Metrology team, is shaping the development of routes to improve the accuracy of cell measurement (the science of cell metrology). In the NML’s Northern Cell Metrology hub, based at Nexus in Leeds, UK, Dr. Rivera is applying metrology to develop improved procedures to detect quantifiable changes typically missed in conventional assays such as subtle morphological drifts are reflective of cellular ageing, phenotypic change that can potentially impact reliability of therapeutic cell products.
For a century, medicine has been transformed through advances in chemistry delivering pharmaceuticals that have been typically stable, with precise doses, and predictable shelf lives. The rise of Advanced Therapy Medicinal Products (ATMPs), gene-modified cells and engineered tissue, has changed the stakes entirely. In this new world, the "drug" is far more complex and in cases a living entity that must be dynamic to deliver an outcome. Ensuring that a batch produced on Tuesday matches one manufactured a month earlier is not just a manufacturing challenge; with our current analytical limitations, it is fundamentally a measurement problem.
The Shape of Health
Traditionally, a "good" cell was defined by its ability to stay alive, yet work from biomanufacturing and measurement-science communities show that viability alone does not guarantee cellular fitness or functional performance. ATMP quality can be defined by Critical Quality Attributes (CQAs); measurable traits that correlate with clinical success and consequently accurate CQA assessment is a critical component of regulatory approval.
Among these, morphology, the size, shape, image intensity and overall structure of the cells has remained an under-leveraged asset. Historically, analysis of morphology has been plagued reproducibility issues due to the subjectivity associated with microscopic observation; one technician’s "healthy" cell is another’s "stressed" outlier. To solve this morphology measurement challenge, Dr. Rivera and her colleagues have explored the potential of label-free imaging technologies such as Deepcell. By using artificial intelligence to interrogate phenotypic signatures without invasive chemical stains, they can identify cell states in their native, physiological condition.
Dr. Rivera (left) and a colleague (right) - photo courtesy of University of Leeds
The Case of the Drifting Factory
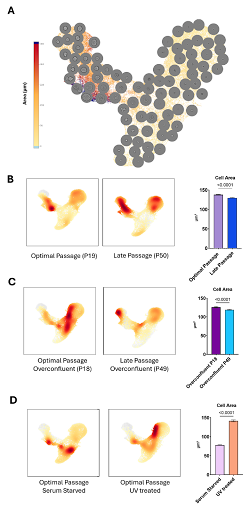
The use of this "digital metrology" is evident in the NML’s study of MRC-5 cells, a line essential for viral vaccine production. These cells have a finite lifespan; they "age" through successive passages, eventually hitting the Hayflick limit and entering senescence—a "zombie" state where they stop dividing but remain metabolically active. Cellular senescence in biomanufacturing of ATMPs pose significant challenges to product quality, consistency, yield and safety. Regulatory and ethical concerns, along with clear clinical guidelines are significant obstacles to defining CQAs in the context of cellular senescence. Using Deepcell’s technology, Dr. Rivera’s team compared early-passage cells against their late-passage counterparts. In addition, serum starvation, UV-induced apoptosis and over-confluence were also assessed to determine morphological differences. While standard viability markers often failed to distinguish between them, the morphological profiling performed using Deepcell REM-i identified a profound "morphological drift". In addition, significant metrology challenges remain, including the need for reference materials, harmonized measurement protocols, and quantitative thresholds that can reliably anchor morphological drift to biological function. Establishing measurement uncertainty, developing cross-platform comparability, and defining the minimum dataset required to qualify morphology-based CQAs represent key next steps for the field. The following features from Deepcell REM-i emerged:
Geometric Precision: Perimeter and area emerged as the most sensitive morphological indicators of cellular ageing. As cells progress toward senescence, their perimeters increase and their shapes become more irregular, followed by a subsequent reduction in overall size as observed in late passage samples.
The Hidden 115: Deepcell REM-i and Human Foundation Modelling identified 115 morphological features, spanning both interpretable measurements and model-derived features. Subtle metrics like "ring width" (measuring the peripheral region of the cell) and "image moment 2" (a measure of cell spread and orientation) provided a candidate measurable threshold for detecting stressed cells long before they would fail a traditional QC check.
Yield at Risk: The data suggests that even minor passage differences can drive morphological drift, directly threatening the yield, potency, and safety of therapeutic products.
Figure 1. (A) Deepcell image‑based single‑cell analysis workflow showing UMAP projection of all captured cells, with representative cell images overlaid according to cluster identity. Cell clusters are distinguished primarily by differences in cell area. (B) UMAP embedding of cells from optimal passage (P19) versus late passage (P50) cultures, demonstrating a significant reduction in mean cell area (µm²) at late passage. Statistical comparisons were performed using Welch’s t-test. (C) UMAP comparison of overconfluent cultures from optimal (P18) and late (P49) passage cells, again revealing a reduction in cell area associated with late passage. Statistical comparisons were performed using Welch’s t-test. (D) UMAP clustering of optimal passage cells subjected to serum starvation or UV‑induced apoptosis, showing the expected differential cell area profiles under these stress conditions. Statistical comparisons were performed using Welch’s t-test. (E) Heatmap distribution of 115 morphological features extracted from Deepcell HFM deep-learning algorithms.
Standardizing the Unstandardizable
The NML’s work contributes to emerging international standardization efforts, including the development of new ISO guidelines under TC276 committee such as ISO/NP 26125 (Tumorigenicity assessment of cellular therapeutic products) and ISO 8934-1 (Requirements for cell viability analytical methods). Defining quantitative thresholds for morphological metrics such as centroid axis, ellipse elongation, and additional model-derived features enables the advancement of real-time, measurement-based monitoring of cellular health.
Picking Winners in the Bioprocess
Deepcell’s technology is migrating rapidly from academic discovery to the production floor. In collaboration with major pharmaceutical partners, Deepcell has analyzed systems ranging from antibody producing workhorses to cutting-edge immunotherapies:
Clonal Selection: In a study of different CHO (Chinese Hamster Ovary) clones, AI identified distinct morphological profiles. Crucially, cells with higher productivity localized to predictive clusters characterized by different morphologies, offering a potential biomarker for manufacturing success and productivity.
The Cell and Gene Therapy Edge: Collaborative research with Dr. Philip Greenberg’s group at Fred Hutchinson Cancer Center analyzed TCR-engineered T cell products. The REM-I platform recognized subtle morphological differences linking them to tumor-killing ability and monitoring of the dynamic balance between activation and exhaustion in real-time.
In advanced therapies, where each manufacturing batch represents substantial clinical and financial value, accurate morphological assessment becomes not merely useful but a potential manufacturing necessity. This can be by both providing a competitive edge through improved process efficiencies and reduced waste as well as routes to provide customer confidence through improvements to quality and safety. Moving from intuition-driven culture practices to measurement-based process control directly supports metrological initiatives such as Consultative Committee for Amounts and Substances CCQM Cell Analysis Working Groups (https://www.bipm.org/en/committees/cc/ccqm/wg/ccqm-cawg) , which prioritises traceability, global comparability, and the development of standards for cell analysis. Recent high-content morphological monitoring studies underscore the need for uncertainty-aware, real-time measures of cell quality. Integrating such metrics into metrological frameworks will reinforce standardisation and improve reproducibility in advanced therapy manufacturing.